2022 AAHA Canine Vaccination Guidelines
Guideline at-a-glance
These Guidelines empower veterinarians to make the best possible personalized recommendations for their patients by determining which vaccines are essential for each individual dog.
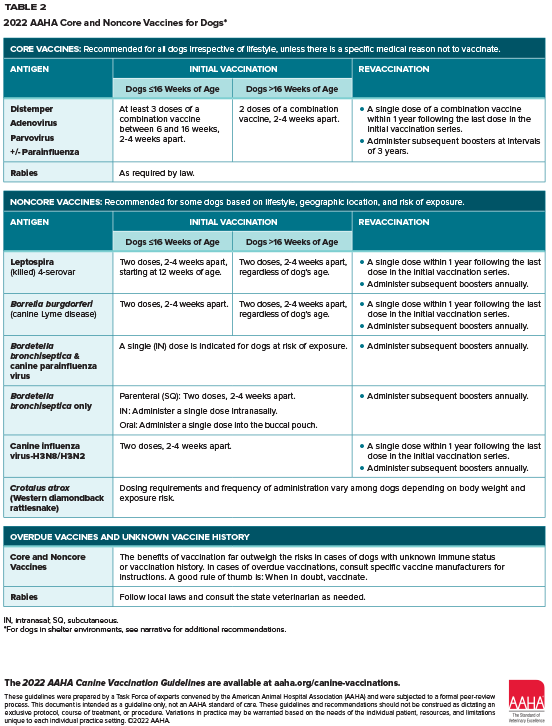
- All dogs should have the following core vaccines (unless there’s a medical reason not to vaccinate):
- Distemper
- Adenovirus
- Parvovirus
- +/- Parainfluenza
- Rabies
- In addition to these, other vaccines are just as essential for some dogs based on their lifestyle and risk. These include:
- Leptospira (should be considered for all dogs based on increasing prevalence)
- Lyme disease
- Bordetella
- Canine influenza
- Rattlesnake toxoid
- For every patient, ask: What should be added to the core vaccines for this dog? Core for an individual patient means they get the vaccines required for ALL dogs PLUS vaccines required based on the dog’s lifestyle and risk factors.
- When vaccines are overdue or unknown, consider that the benefits of vaccinating outweigh the risks in most cases. A good rule of thumb is: When in doubt, vaccinate.
- Train your team to talk to clients about vaccines and why they are a vital part of their dog’s health plan.
Watch this summary of the 2022 AAHA Canine Vaccination Guidelines
Abstract
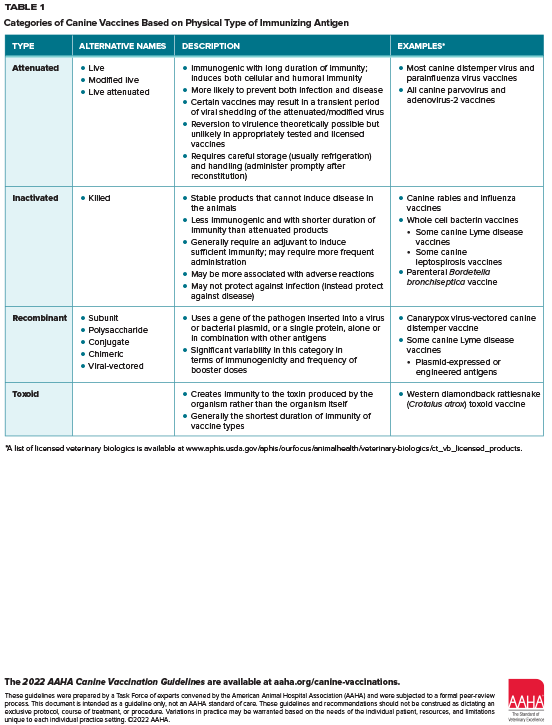
These guidelines are an update and extension of previous AAHA peer-reviewed canine vaccination guidelines published in 2017. Vaccination is a cornerstone of canine preventive healthcare and one of the most cost-effective ways of maintaining a dog’s health, longevity, and quality of life. Canine vaccination also serves a public health function by forming a barrier against several zoonotic diseases affecting dogs and humans. Canine vaccines are broadly categorized as containing core and noncore immunizing antigens, with administration recommendations based on assessment of individual patient risk factors. The guidelines include a comprehensive table listing canine core and noncore vaccines and a recommended vaccination and revaccination schedule for each vaccine. The guidelines explain the relevance of different vaccine formulations, including those containing modified-live virus, inactivated, and recombinant immunizing agents. Factors that potentially affect vaccine efficacy are addressed, including the patient’s prevaccination immune status and vaccine duration of immunity. Because animal shelters are one of the most challenging environments for prevention and control of infectious diseases, the guidelines also provide recommendations for vaccination of dogs presented at or housed in animal shelters, including the appropriate response to an infectious disease outbreak in the shelter setting. The guidelines explain how practitioners can interpret a patient’s serological status, including maternally derived antibody titers, as indicators of immune status and suitability for vaccination. Other topics covered include factors associated with postvaccination adverse events, vaccine storage and handling to preserve product efficacy, interpreting product labeling to ensure proper vaccine use, and using client education and healthcare team training to raise awareness of the importance of vaccinations.( J AmAnimHosp Assoc 2022; 58:1–19. DOI 10.5326/JAAHA-MS-Canine Vaccination Guidelines)